The growing interest in PEEK-OPTIMA™ spinal implants in China received further impetus at the 11th Congress of the Chinese Association of Orthopaedic Surgeons (CAOS) held in partnership with the North American Spine Society (NASS). At this event, Fule Science & Technology Development, Beijing, and Invibio Biomaterial Solutions partnered to demonstrate the new Uplifter™ Expandable Anterior Lumbar Interbody Fusion Device in a hands-on workshop in Shanghai.
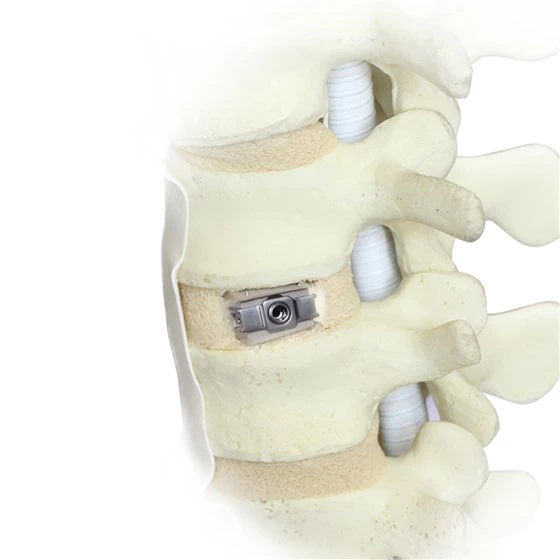
The Uplifter is the first domestic expandable hybrid cage, whose constituent materials are Invibio’s PEEK-OPTIMA™ Natural polymer and titanium. Indications for use of the Uplifter comprise Degenerative Disc Disease (DDD) of L2-L5, nerve root-canal stenosis, and lumbar spondylolisthesis.
• Minimized impaction contributing to preservation of endplate integrity
• Controlled expansion and anatomical fit helping to maximize indirect decompression through disc height restoration
• Continuous expansion that can maintain stability by optimizing the fit
Professor Renfu Quan, a faculty member at Hangzhou Xiaoshan TCM hospital responsible for on-site cadaver workshops, explained the standard surgical techniques for OLIF and demonstrated the procedures for the surgeons present in a lab setting. Using Fule’s retractors and instruments, he showed how to implant their expandable”Poster“ cage.
This live demo was available to surgeons who attended the workshop in Shanghai and was also broadcast directly to the CAOS/NASS congress in Hangzhou. The demo has also been published through the CAOS online education platform, being available to more than 10,000 participants in the CAOS congress.
Radiolucent biomaterial allows better fusion assessment
“PEEK has been used for interbody fusion devices for many years. Both, the literature and my personal experience indicate that PEEK provides better clinical outcomes compared with titanium-only approaches in spinal fusion. There is a higher fusion rate and lower subsidence”, Dr. Xifeng Zhang, from Beijing 301 Hospital, emphasized in his ‘Review of Clinical Applications and results associated with Direct-Lateral Lumbar Interbody Fusion (DLIF). Indeed, the potential benefits of PEEK-OPTIMA Natural in conventional spinal cages may translate to expandable cages. They include radiolucency, with no artifacts present, allowing better fusion assessment by CT and MRI and a modulus of elasticity close to that of bone, reducing stress shielding, which may otherwise contribute to implant subsidence.“
Guoping Fan, Technology Director at Fule Science & Technology Development Company commented: “To drive advances in orthopedics we partner with a variety of experts ranging from material and technology experts to orthopedic surgeons treating patients. The in-depth technical cooperation with Invibio for example allowed the development and introduction of this first hybrid PEEK-titanium expandable cage in China for DLIF and OLIF procedures. Now we are working full speed to develop the spinal fusion devices with PEEK-OPTIMA HA Enhanced.”
In China, Invibio´s enhanced PEEK is a relatively new thermoplastic biomaterial offering all of the clinical advantages of the unfilled “PEEK-OPTIMA Natural” polymer. In addition, it has been shown to improve bone apposition compared to unfilled PEEK-OPTIMA Natural in a pre-clinical ovine model at 4 weeks and 12 weeks(2), and also demonstrated performance advantages in a pre-clinical cervical spine fusion model in sheep at 6 and 12 weeks.(2) In PEEK-OPTIMA HA Enhanced, Hydroxyapatite (HA), a well-known osteoconductive material, is fully integrated into the polymer matrix, and not simply coated on the surface. Early clinical results for cervical and lumbar spinal fusion reveal potential patient benefits and specific improvements for the enhanced biomaterial including solid fusions as early as 6 months, beneficial clinical outcomes at early time points, improvements in overall pain and neurological function.(3)
With China's accelerating interest in innovative materials such as PEEK-OPTIMA from Invibio for spinal implants, it is expected that more and more patients can potentially benefit from implants using the high performance polymer. Today, more than 140 devices in China have been approved by the CFDA [China Food and Drug Administration, aka SFDA (State Food Drug Administration) ], with over 10 years of accumulated clinical history in China.(4)
The 11th CAOS/NASS congress in May marks the second time Invibio has partnered with a medical-device manufacturer in China to support surgeon education. The company´s PEEK-OPTIMA implants now have over 15 years of clinical history and approximately nine million PEEK-OPTIMA devices have been implanted worldwide.(5)
References:
(1) PEEK” is the abbreviation for polyetheretherketone, a high-performance polymer from the PAEK (polyaryletherketone) family.
(2) Walsh WR, Pelletier MH, Bertollo N, Christou C, Tan C. Does PEEK/HA Enhance Bone Formation Compared With PEEK in a Sheep Cervical Fusion Model? Clin Orthop Relat Res. 2016 Nov; 474(11):2364-2372.
(3) Testimonials presented have been provided by practicing orthopaedic surgeons. Their view and experience are their own and do not necessarily reflect those of others. “Invibio” disclaims any liabilities or loss in connection with the information herein.
(4) Data on file at Invibio Biomaterial Solutions.
(5) Data on file at Invibio Biomaterial Solutions.
Further information is available on https://invibio.com
About Fule Science & Technology Development
Established in 1994, Beijing Fule Science & Technology Development Co. Ltd is a manufacturer and trader specialized in the research, development and production of Orthopaedic products including spinal products, trauma products, multi-functional external fixator, knee meniscus suturing device, rehabilitation instrument and related instruments. The company's products have been widely used by thousands of large and medium-sized hospitals and achieved a good report, winning a high reputation from many orthopedic experts. Further information is available on http://www.fulekeji.com/en/index.php