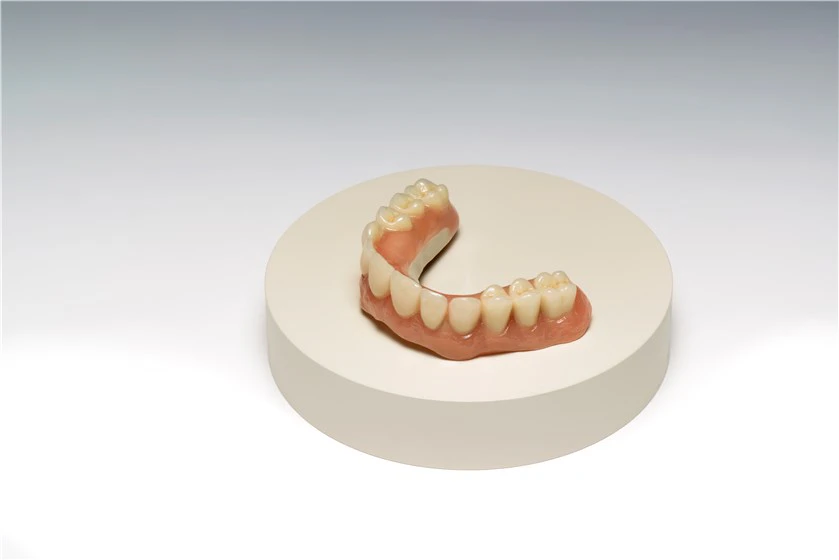
JUVORA™ solutions are to become available to more dentists and their patients worldwide: Straumann, a global leader in implant, restorative and regenerative dentistry, has added the JUVORA dental disc (“JUVORA”) to its portfolio. The company will provide milled JUVORA frameworks and also distribute the dental disc as part of the Straumann® CARES® digital solutions system. The dental disc can contribute to an improved quality of life for patients. It is suitable for fixed and removable prosthetics, such as implant dentures, full and partial dentures and overdentures, crowns and bridges.
Straumann CARES digital solutions are facilitating dentistry's migration to digital-design techniques and workflows. The move to combining this with high-performance polymers is providing new digital production and treatment options for dentists and patients. Interest in the use of these materials for restorative dentistry is growing, as indicated in over 70 peer-reviewed publications over the last two years, as well as an increasing number of clinical studies and patient surveys.
Strong clinical data, real-world patient experience, and backing from leaders within the dental profession are expected to support increasing use of this innovative product. In addition, the high level of service provided by Juvora Limited includes regulatory clearance/certification and a JUVORA training guide. The training guide is available as an APP via app stores and includes training and product information to support dental labs and dentists.
JUVORA prosthetics are made entirely of implantable PEEK-OPTIMA™ polymer from Invibio Biomaterial Solutions. This innovative material flexes and, unlike metal, enables the absorption of shock in the masticatory system, protecting the device and patient, especially in cases of bruxism which can lead to parafunction.(2) In addition it offers the potential for the CAD/CAM milling of JUVORA-based solutions up to three times faster than an equivalent titanium product.(3) Today, dental patients are benefiting from a JUVORA prosthetic, which seeks to improve the quality of life for patients, as well as clinical outcomes for dentists(4), and offers manufacturing efficiency(3) as an added benefit. In a patient survey over 20 months, 99% of patients rated JUVORA highly for comfort.(5)
Paving the way to improving the quality of life of dental patients
John Devine, Medical Business Director at Invibio Biomaterial Solutions, comments, “For many years, Straumann has led innovation in dentistry and dental biomaterials. Working together with them as one of the industry's leaders will further pave the way to digital dentistry and allow patients across the globe to access customized high quality prosthetic dental implants based on our PEEK biomaterials.”
Since PEEK-OPTIMA Natural was launched as the world’s first implantable PEEK polymer more than 15 years ago, Invibio and Juvora have pioneered the use of high-performance polymers in long-term implantable medical-device applications such as Spine, Arthroscopy, Trauma and Dental. In the process, evidence of the clinical benefits of JUVORA has been amassed as a framework material in implant and tooth-borne prosthetics. European CE mark certification for JUVORA was secured in 2012, followed by initial US FDA approval in 2014, and a further FDA approval in 2017 for JUVORA in implant-borne restorations. In 2017, JUVORA was also granted a US patent.
(1) “PEEK” is the abbreviation for polyetheretherketone, a high-performance polymer from the PAEK (polyaryletherketone) family.
(2) M Rosentritt, S. Schneider-Feyrer, M. Behr, V. Preis (2017). in Vitro Shock Absorption Tests on Implant-Supported Crowns: Influence of Crown Materials and Luting Agents. The International Journal of Oral & Maxillofacial Implants, 2017 May 18. doi: 10.11607/jomi.5463.
(3) Invibio data on file. Independent laboratory test indicated milling JUVORA implant prosthetic framework in one hour, 21 minutes, versus Titanium equivalent in three hours, 50 minutes.
(4) Siewert, B. (2017), PEEK in Dental Prosthetics. Industry Symposium. EAO Conference. Madrid, Spain. 5th October 2017. Available to watch at https://www.youtube.com/watch?v=wNvSwG4CaeYB.
(5) Invibio data on file. JUVORA prosthetics patient survey from 92 cases, July 2013-March 2015.
Further information is available on www.juvoradental.com
About Juvora
Juvora Limited is the legal manufacturer of the JUVORA dental disc and alongside Invibio (see below) is part of the Victrex plc group of companies (see below). The JUVORA solution can be used as a metal alternative for long-term implant borne, fixed and removable prosthetic frameworks made with precision through CAD/CAM workflows. It is made from Invibio Biomaterial Solutions´ PEEK-OPTIMA high performance polymer. Find out more at www.juvoradental.com
About Invibio Biomaterial Solutions
Invibio, part of the Victrex plc group of companies, is a global leader in providing high-performance biomaterial solutions to medical device manufacturers. The company provides PEEK-OPTIMA™ polymers, advanced technical research and support and manufacturing of components for spine, trauma and orthopaedic and dental medical segments for the development of long-term implantable medical devices. Today, Invibio’s PEEK-OPTIMA™ polymers are used in approximately 9 million implanted devices worldwide.
JUVORA™, INVIBIO™, PEEK-OPTIMA™, INVIBIO BIOMATERIAL SOLUTIONS™ are trademarks of Victrex plc or its group companies. All rights reserved. Find out more at https://invibio.com/